Analysis of Vacuum Distillation in Metallurgy
Vacuum distillation in metallurgy is mainly reflected in the vacuum smelting of non-ferrous metal materials, with a short process, less consumption, higher metal recovery, and many advantages, like non-waste (waste water, waste residue, waste gas), no pollution to the environment, little processing costs, etc. Therefore, vacuum distillation has been more and more used in non-ferrous metal refining.
The basic principles of metal vacuum distillation are divided into: metal evaporation, separation of crude metal and alloy components distillation, metal vapor condensation.
Taking the vacuum distillation of lead and its alloys as an example, firstly, the separation of lead and zinc is based on the poor mutual dissolution ability of the two metals, dissolving about 2% of zinc in lead above 417.66℃, and about 0.9% of lead in zinc. Once exceeds these quantities, they two apart into two layers. Due to the difference of 9.21X104 between the gaseous and liquid zinc / lead fingers, the two metals were quite completely separated by vacuum distillation. The industrial vacuum dezincification equipment designed by Isbell in 1947 is shown in the figure below:
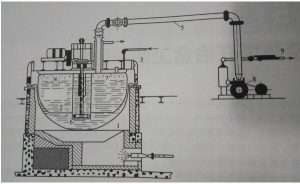
1.Lead pot 2. Bell jar 3. Condensation structure 4. Water jacket 5. Vacuum line 6. Agitator 7. Condensed zinc 8. Vacuum pump 9. Exhaust
At the beginning, the vacuum degree is about 0.5mmHg, after 1.5-2 hours of vacuum distillation, the lead in the zinc has been greatly reduced, then the vacuum rises to 0.05mmHg, the lead content of zinc reduced to 0.01% to 0.05%. Recycled 80% to 90% of zinc metal.
By different forms of vacuum distillation, it can be applied to lead silver alloy, tin alloy, arsenic, selenium and tellurium alloy and so on. For specific application and selection of vacuum system, please consult EVP engineers team.
Tags: vacuum unit, vacuum system, Vacuum Distillation, Metallurgy
Edited by: Emily Fu / EVP Vacuum Solutions

